This ‘Elektron-V’ system could produce up to 1900
liters per day, with an average power load of 860 watts. Its mass was 150
kg. The design lifetime was 3 years and the First unit operated for three
and a half years.
WHERE THE ‘ELEKTRON’ IS BUILT
The system was developed and fabricated by a corporation called
Niichimmash, also transliterated NIIKHIMMASH. In English, this means
“Scientific-Experimental Institute of ChemicalMachine-Building,”
in Sergiyev Posad (Zagorsk), near Moscow. It also builds chemical
rocket engines and other chemistry-related products, including for
the Russian military.
In 1992, visiting NASA engineers were shown an Elektron
unit that weighed 110 kg and expended 3385 watt-hours per lb of
oxygen (a comparable US unit made by Hamilton Standard, the
‘SPE’, uses 2400 watt-hours per lb of O2). The unit
used a circulating KOH electrolyte, and the hydrogen and oxygen
product gases are separated from the water using ‘static separators’.
|
 |
The unit was in a titanium housing with a nitrogen blanket. The oxygen
is not stored but is discharged directly into the cabin at 45 psi. Hydrogen
is vented overboard although there was hardware to divert it to a planned
Sabatier Reactor for CO2 reduction (never installed).
Niichimmash had one Elektron unit without pressure shell, for classroom
training, and two flight units, one that had been operating since 1985
to “lead” the Mir flight unit, and the other available for
mission support and off-nominal tests. Another flight unit was at Star
City for cosmonaut training.
TECHNICAL SPECIFICATIONS
In the September 1993 3rd Edition of “Soviet Space Stations As Analogs”,
B.J. Bluth and Dennis Fielder, produced by Grumman Aerospace in Reston,
Va., section 1.4.7.3.4 (p. I-238) contains technical data on the
electrolysis reaction. Electrolysis of pure water at normal temperatures
is enhanced by the presence of an alkali to support ionization and increase
conductivity:
KOH = K+ + OH-
Maximum electrolyte conductivity can be achieved with its concentration
at about 30% KOH. At the anode, negative ions of the hydroxyl group OH-
migrate towards the positive anode and discharge, releasing electrons
4 OH- - 4e = 2H2O + O2 , releasing oxygen and forming water, while water
molecules combine with the hydroxyl molecules, migrate to the cathode,
oxidize, and release hydrogen thusly:
2H2O + 2e = 2 OH- + H2
According to this document, the Mir electrolysis system had a mass of
140 kg, had twelve cells each with a cell voltage of 2v and a size of
0.8m x 0.13m, used 800 w with max current at 2V of 400 amp.
NASA TRAINING MANUAL ON ‘ELEKTRON’
From the “NASA Familiarization Manual on Russian Segment Crew Systems”,
Published in 1997.
3.1. Oxygen Supply System
The oxygen supply system of the Russian segment consists of the Elektron
unit, two solid fuel oxygen generators (TGK), and deliverable oxygen from
the Progress cargo spacecraft.
The Elektron unit is the prime source of oxygen and operates on the principle
of the electrochemical decomposition of water. The TGK and the Progress
deliverable oxygen are auxiliary sources of oxygen.
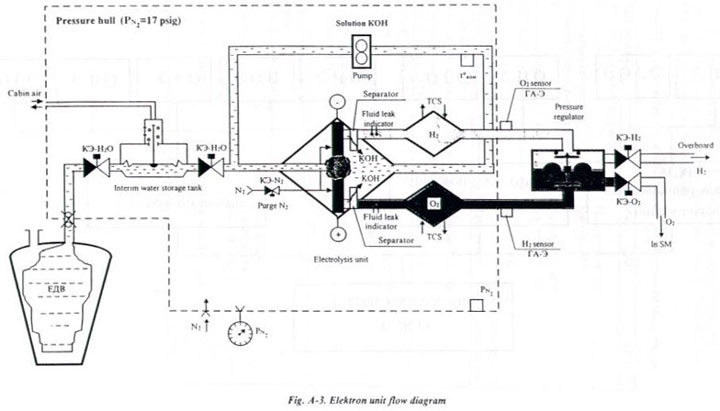
The Electron consist of a liquid loop with 30-percent potassium hydroxide
(KOH) in solution, gas lines with a pressure regulator, and electromagnetic
valves (KEh). The liquid loop is placed into a pressure-tight hull pressurized
with nitrogen for safety reasons. External to the pressure-tight hull,
the liquid loop is connected to a water tank (YeDV), which is replaced
periodically.
The Elektron unit is installed behind
panels 429 and 430 in the SM.
The liquid loop includes an electrolysis unit, two heat exchangers, a
pump, and an internal water storage tank. The pump provides the circulation
of the electrolyte through the electrolysis unit. The internal water storage
tank acts as a membrane pump to replenish the amount of water decomposed
into oxygen and hydrogen.
The pressure regulator maintains a preset pressure differential in the
oxygen and hydrogen lines.
Fig. A-3 shows a diagram
of the electrolysis process. Oxygen is generated in the system by
the electrolysis of the water content in potassium hydroxide solution.
Water is made up of 89% oxygen by weight. The reaction breaks down water
into its constituents of oxygen and hydrogen. The oxygen is released directly
into the atmosphere of the SM and the hydrogen is vented to vacuum. The
electrolysis unit is made up of 12 electrolysis cells which are enclosed
in a blastproof housing. The cells are cooled by the thermal control system
(STR). The decomposition of 1 kg (2.2 lbs) of water yields 25 L (0.88
ft3) of oxygen per hour at a pressure of 760 mmHg, which is enough to
support one crew member for one day. To provide the daily amount of oxygen
for 3 - 4 crew members, 3 - 4 kg (6.6 - 8.8 lbs) of water must be decomposed.
Power consumption of the process is ~ 1 kW.
The Elektron unit is controlled by the on-board computer system. The
following parameters are monitored in the process of operation: valve status,
oxygen and hydrogen pressure internal to pressure hull of the Elektron,
hydrogen concentration in the oxygen line, and oxygen concentration
in the hydrogen line.
If the hydrogen pressure in the oxygen line reaches 2% of the total line
pressure, the signal “GA-Eh srabotal” (GA-Eh response to maximum
value) is transmitted to the Integrated Control Panel (InPU) (Fig. B-I).
A similar signal is generated if the oxygen pressure in the hydrogen
line reaches 2% of the total line pressure.
Another signal “Prover elektron” (Test Elektron) is transmitted
to the Integrated Control Panel (InPU) if any of the following conditions
occur:
• temperature of the electrolyte reaches 65ºC
(150ºF)
• oxygen pressure in line exceeds 0.65 kg/cm2
(9.5 psig)
• pressure of the interim water storage tank drops
below 900 mm H20
• nitrogen pressure in the pressurized hull
drops to 0.9 kg/cm2 (13.3 psig)
• differential pressure between
oxygen and hydrogen lines exceeds 500 mmH2O
• electrolyte
is detected in the hydrogen or oxygen lines.
For either of the signals described above, the Elektron will perform an
automatic shutdown
|




